|
In creating even more unique arrangements of amino acids, even more types of interactions are formed. Our discussion of secondary structure has introduced us to the carbonyl and amino groups (that are present in all amino acids) and to the hydrogen bond. The strands in a beta pleated sheet can have either the same directionality or alternating directionality, creating parallel and antiparallel beta sheets- respectively. Beta Sheetsīeta sheets are strands of amino acids that are in line with each other, creating a large sheet of connected amino acids. Hydrogen bonds are made from the carbonyl oxygen, and the amide group four positions away (not depicted). Alpha HelixĪn alpha helix is a strand of amino acids arranged in a coil, much like the coil of DNA. Both of these structures contain hydrogen bonds, but differ in where the hydrogen bonds are formed. The secondary structure of a protein describes key features of a folded protein: the alpha helix and the beta pleated sheet. When amino acids in a protein’s sequence are changed, it is possible for the protein to lose or change its shape. 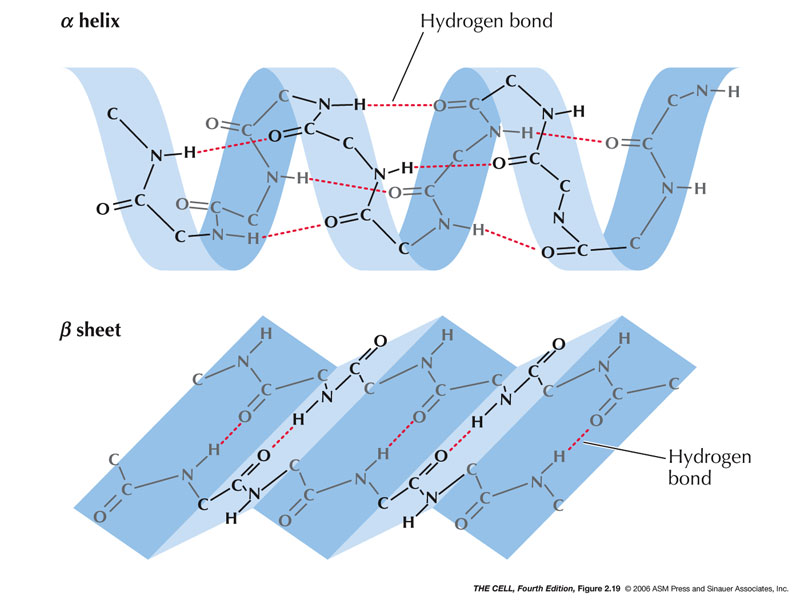
The primary sequence of a protein can affect all other levels of protein structure. 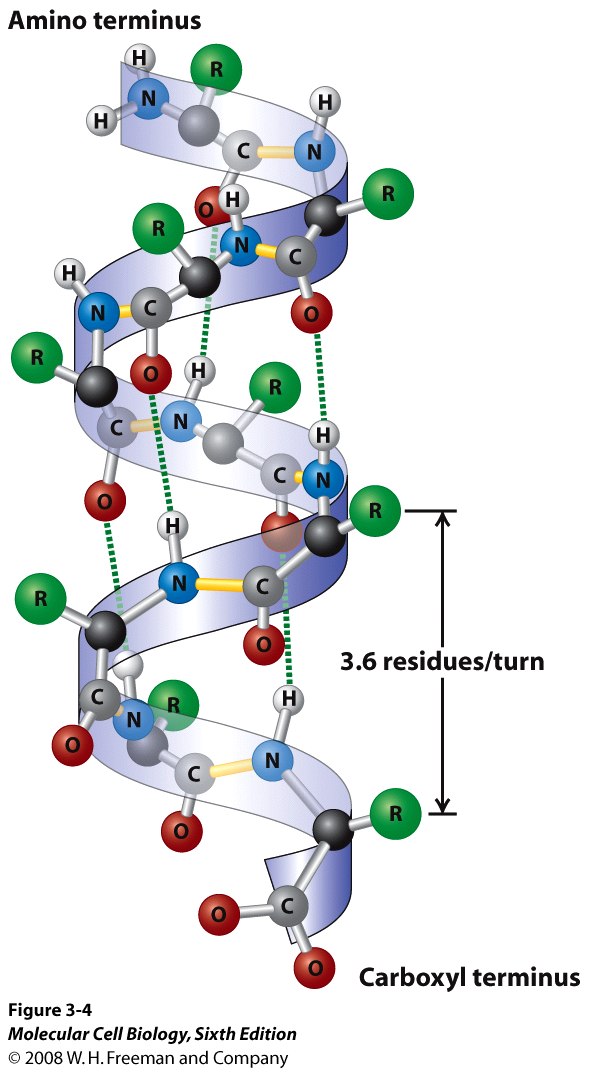
The primary level of protein structure, although the most basic, is vital to the shape the protein will take on. Quaternary Structure describes how multiple protein chains interact with each other. how secondary structures and unstructured regions of the protein interact. Tertiary structure more broadly describes how the protein folds – i.e. Secondary Structure describes the formation of two critical configurations: the alpha helix and beta pleated sheet. The most basic being primary structure, which describes a chain of amino acids. Describing Protein Folding through Structure Levels of Protein Structureįour levels of protein structure describe proteins. The shape that a protein takes on is vitally important to how it behaves. For example the protein hemoglobin takes on a specific shape that allows it to carry oxygen through the body, and the protein collagen has a different but still important structure that allows skin to maintain its elasticity. In many biology courses, it is often said structure=function, and proteins are no exception. Protein folding is the process in which a protein takes on its shape. Protein Folding is Important to Protein Function You will also learn how a protein folds, explained along side the four levels of protein structure. 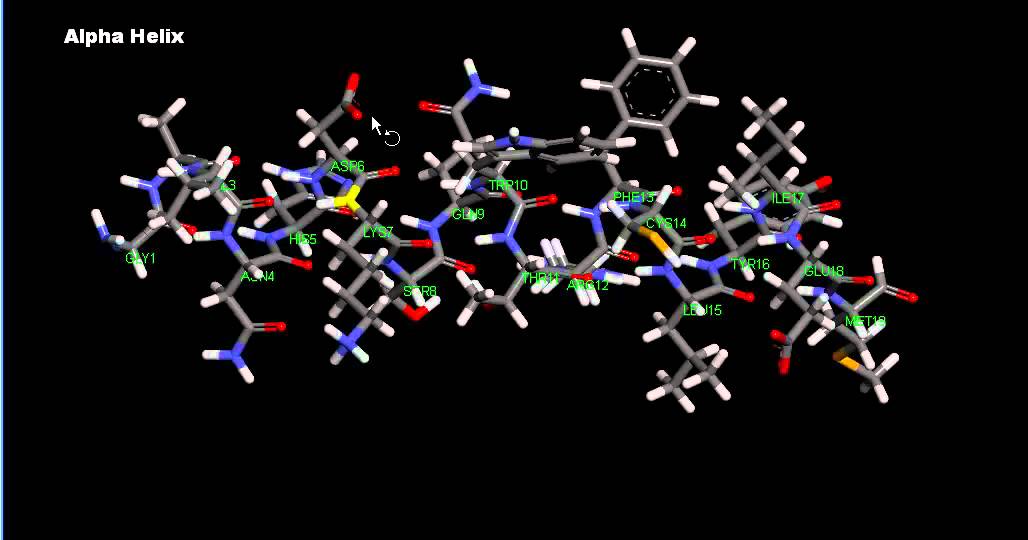
Alpha helices are common in important biological proteins such as keratin (skin), hemoglobin (blood) & myoglobin (muscle)ĥ0mm bonds with white inserts represent hydrogen atoms that form hydrogen bonds.In this tutorial, you will learn why protein folding is important to how a protein functions. The "R" groups can be augmented with additional parts to show the most commonly occurring amino acid residues methionine, alanine, leucine, glutamate and lysine in the alpha helix polypeptide chain. It fits the major groove easily & shows steric hindrance compared to the minor groove. the major groove of our 12 base pair DNA double helix model. The “R” groups are indicated with a univalent green atom & face outward where interact with other structures, e.g. This helical polypeptide backbone consists solely of alpha carbons and amino & carboxyl groups. It shows protein secondary structure based on 3.6 amino acid residues per turn. This Indigo® alpha helix model consists of 20 amino acid cores that form a right handed or clockwise spiral.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
